 
The results obtained from scanning electron spectroscopy, X-ray photoelectron spectroscopy, X-ray diffraction, thermal gravimetric analysis, and molecular energy level calculations show that PES favors the formation of a stable solid electrolyte interphase, not only on the anode but also on the cathode of the LiMn 2O 4/graphite battery, effectively preventing electrolyte decomposition. As such, cathode is observed to be more sensitive to electrolyte environment. The electrons flow through an external circuit and the hydrogen ions selectively move across the PEM to the cathode. Water reacts at the anode to form oxygen and positively charged hydrogen ions (protons). The electrolyte is aqueous copper(II) sulfate solution.Because. The influence of electrolyte on cathode is much more obvious than that on anode: 85.9 and 445 increase in failure strain, 8 and 12.7 drop in strength of anode and cathode respectively with electrolyte. In a polymer electrolyte membrane (PEM) electrolyzer, the electrolyte is a solid specialty plastic material. The capacity retention is 91% and the swell value in thickness is 3.4% for the cell with PES after 150 cycles at 60 ☌, while the respective values were 68% and 36.4% for the cell without additive, and 82% and 9.1% for the cell with VC. An impure strip of copper acts as the anode and a thin pure strip of copper acts as the cathode. It was found that the cell based on the developed electrolyte exhibits better cyclability and exhibits better dimensional stability at elevated temperatures. 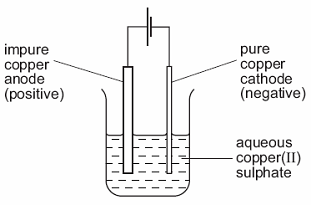
The cycling performance of the LiMn 2O 4/graphite cell, based on the developed electrolyte at 60 ☌, was evaluated by a constant current charge/discharge test, with comparison of the electrolyte using vinylene carbonate (VC) as additive. Commonly used electrolytes in electrolytic cells include water (containing dissolved ions) and molten sodium. To improve the cyclability of a LiMn 2O 4/graphite lithium ion battery at elevated temperature, a carbonate-based electrolyte using prop-1-ene-1,3-sultone (PES) as additive was developed. Cathode (which is negatively charged for electrolytic cells) Anode (which is positively charged for electrolytic cells) Electrolyte The electrolyte provides the medium for the exchange of electrons between the cathode and the anode.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
